Compare and Contrast Alpha Beta and Gamma Radiation Chemistry Semester Course Review
Radiations is energy. It can come up from unstable atoms that undergo radioactive decay, or it can be produced by machines. Radiation travels from its source in the form of energy waves or energized particles. There are different forms of radiation and they take dissimilar backdrop and effects.
On this page:
- Ionizing and not-ionizing radiation
- Electromagnetic spectrum
- Types of ionizing radiation
- Periodic Table
Non-Ionizing and Ionizing Radiation
There are 2 kinds of radiations: non-ionizing radiation and ionizing radiation.
Not-ionizing radiations has enough energy to move atoms in a molecule around or crusade them to vibrate, merely not plenty to remove electrons from atoms. Examples of this kind of radiation are radio waves, visible light and microwaves.
Ionizing radiation has so much energy it tin knock electrons out of atoms, a procedure known as ionization. Ionizing radiation can bear upon the atoms in living things, and so it poses a wellness chance past damaging tissue and DNA in genes. Ionizing radiation comes from x-ray machines, cosmic particles from outer infinite and radioactive elements. Radioactive elements emit ionizing radiation every bit their atoms undergo radioactive decay.
Radioactivity is the emission of energy in the grade of ionizing radiationRadiation with and then much energy it tin knock electrons out of atoms. Ionizing radiation can affect the atoms in living things, so information technology poses a health chance past damaging tissue and DNA in genes. . The ionizing radiations that is emitted can include alpha particlesA grade of particulate ionizing radiations made up of ii neutrons and ii protons. Alpha particles pose no direct or external radiation threat; however, they tin can pose a serious health threat if ingested or inhaled. , beta particlesA form of particulate ionizing radiation made upwards of pocket-sized, fast-moving particles. Some beta particles are capable of penetrating the skin and causing damage such as skin burns. Beta-emitters are most chancy when they are inhaled or swallowed. and/or gamma raysA class of ionizing radiation that is made up of weightless packets of free energy called photons. Gamma rays can pass completely through the homo trunk; as they pass through, they can cause impairment to tissue and Dna. . Radioactive decay occurs in unstable atoms called radionuclides.
Electromagnetic Spectrum
The energy of the radiations shown on the spectrum beneath increases from left to correct as the frequency rises.
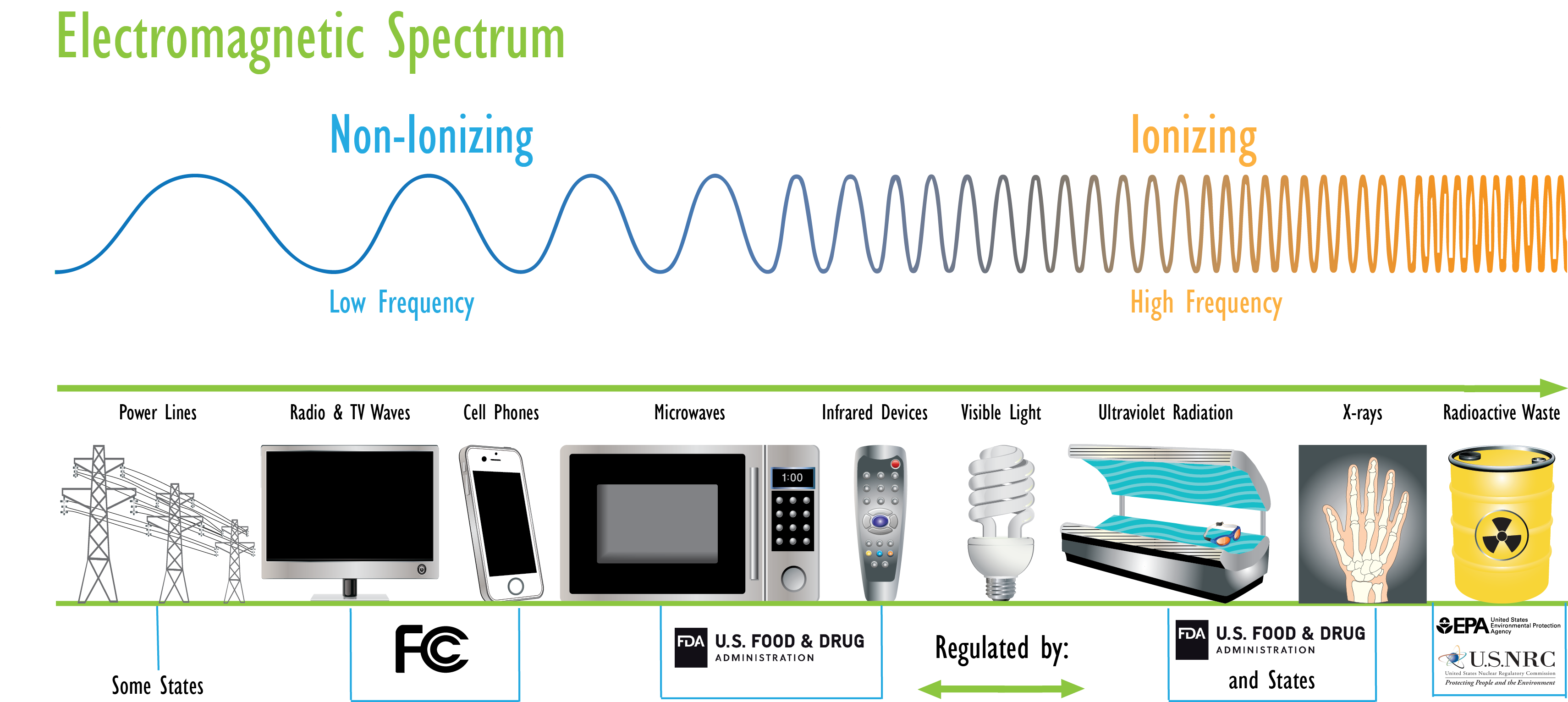
EPA'southward mission in radiation protection is to protect homo health and the environment from the ionizing radiation that comes from human being use of radioactive elements. Other agencies regulate the non-ionizing radiation that is emitted past electric devices such every bit radio transmitters or cell phones (See: Radiation Resource Outside of EPA).
Types of Ionizing Radiations
Alpha Particles
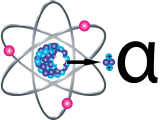
Alpha particles (α) are positively charged and made up of ii protons and ii neutrons from the cantlet's nucleus. Blastoff particles come up from the disuse of the heaviest radioactive elements, such every bit uranium, radium and polonium. Even though alpha particles are very energetic, they are and then heavy that they use upwardly their free energy over short distances and are unable to travel very far from the atom.
The health effect from exposure to alpha particles depends profoundly on how a person is exposed. Alpha particles lack the free energy to penetrate even the outer layer of skin, then exposure to the outside of the body is non a major concern. Inside the body, however, they tin exist very harmful. If alpha-emitters are inhaled, swallowed, or get into the trunk through a cutting, the alpha particles tin harm sensitive living tissue. The way these large, heavy particles crusade damage makes them more than dangerous than other types of radiation. The ionizations they cause are very close together - they can release all their energy in a few cells. This results in more astringent damage to cells and DNA.
Beta Particles
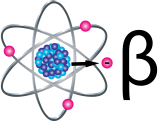
Beta particles (β) are small, fast-moving particles with a negative electrical charge that are emitted from an atom'south nucleus during radioactive decay. These particles are emitted by certain unstable atoms such as hydrogen-3 (tritium), carbon-14 and strontium-xc.
Beta particles are more penetrating than alpha particles, but are less damaging to living tissue and DNA because the ionizations they produce are more than widely spaced. They travel farther in air than blastoff particles, but can exist stopped by a layer of clothing or past a thin layer of a substance such as aluminum. Some beta particles are capable of penetrating the skin and causing damage such as skin burns. All the same, as with alpha-emitters, beta-emitters are about hazardous when they are inhaled or swallowed.
Gamma Rays
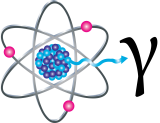
Gamma rays (γ) are weightless packets of energy called photons. Unlike alpha and beta particles, which have both free energy and mass, gamma rays are pure energy. Gamma rays are similar to visible light, but have much college free energy. Gamma rays are frequently emitted along with alpha or beta particles during radioactivity.
Gamma rays are a radiation adventure for the entire torso. They can easily penetrate barriers that tin can stop blastoff and beta particles, such equally peel and habiliment. Gamma rays accept so much penetrating power that several inches of a dense material like lead, or even a few anxiety of concrete may exist required to stop them. Gamma rays can pass completely through the human trunk; as they pass through, they can crusade ionizations that impairment tissue and DNA.
X-Rays
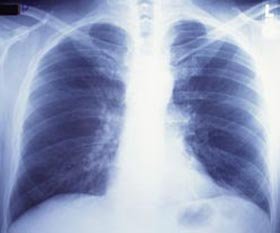
Considering of their use in medicine, almost everyone has heard of x-rays. 10-rays are similar to gamma rays in that they are photons of pure energy. X-rays and gamma rays have the same bones properties just come from unlike parts of the atom. X-rays are emitted from processes outside the nucleus, but gamma rays originate inside the nucleus. They as well are generally lower in energy and, therefore less penetrating than gamma rays. 10-rays can be produced naturally or past machines using electricity.
Literally thousands of x-ray machines are used daily in medicine. Computerized tomography, commonly known as a CT or Cat scan, uses special x-ray equipment to make detailed images of basic and soft tissue in the torso. Medical 10-rays are the single largest source of man-fabricated radiation exposure. Learn more than most radiation sources and doses. X-rays are also used in manufacture for inspections and procedure controls.
Periodic Table
Elements in the periodic table tin can have on several forms. Some of these forms are stable; other forms are unstable. Typically, the virtually stable form of an chemical element is the most common in nature. However, all elements take an unstable form. Unstable forms emit ionizing radiations and are radioactive. There are some elements with no stable form that are always radioactive, such every bit uranium. Elements that emit ionizing radiations are called radionuclides.
Source: https://www.epa.gov/radiation/radiation-basics
0 Response to "Compare and Contrast Alpha Beta and Gamma Radiation Chemistry Semester Course Review"
Post a Comment